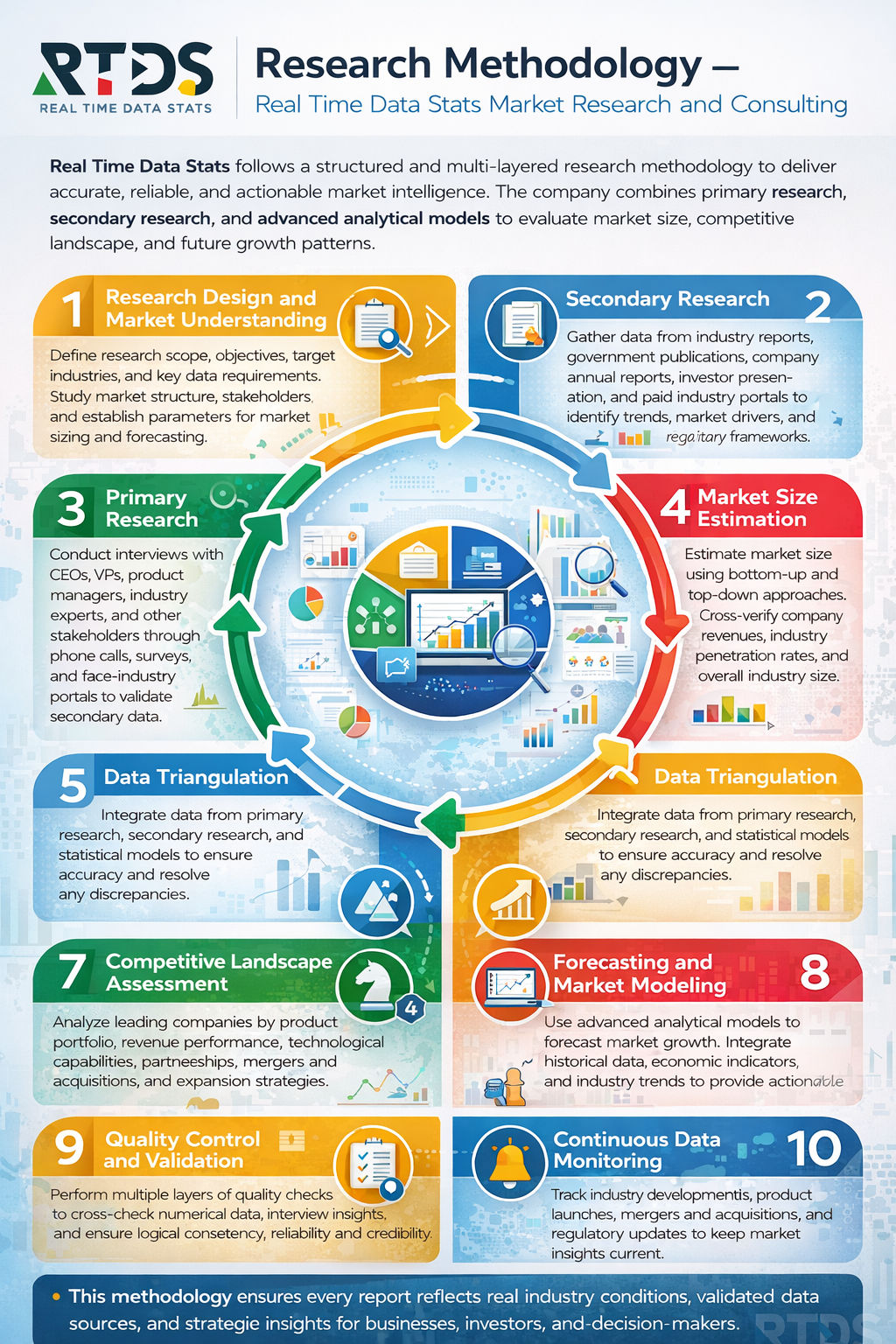
According to insights from Real Time Data Stats, the Synthetic Protein Circuits Market was valued at USD 0.50 billion in 2025. It is expected to grow from USD 0.52 billion in 2026 to USD 1.40 billion by 2033, registering a CAGR of 16% during the forecast period (2026–2033).
MARKET SIZE AND SHARE
The global synthetic protein circuits market is expanding fast, driven by rising demand in advanced therapeutics and precision diagnostics. Leading biotechnology firms and academic spin-offs in North America and Europe hold over 60% of revenue, focusing on programmable cell therapies, gene regulation systems, and next-generation biologics. Strong research funding, strategic partnerships, and rapid clinical translation continue to strengthen their dominance and accelerate innovation across this evolving field.
Market share will shift as the technology matures and adoption widens across industries. Asia-Pacific players are set to gain strong ground by 2032, increasing competition and regional balance. Therapeutic applications, especially in cancer and metabolic disorders, will lead revenue generation, while biosensing and biomanufacturing segments will grow rapidly. This shift will diversify revenue streams, reduce early-stage concentration, and open new commercial pathways globally.
INDUSTRY OVERVIEW AND STRATEGY
The synthetic protein circuits industry merges synthetic biology with protein engineering to create programmable molecular systems that control cellular behavior. It is a highly interdisciplinary and R&D-intensive field, poised to revolutionize precision medicine, advanced diagnostics, and sustainable bioproduction. The industry is currently in a translational phase, moving from foundational academic research toward commercial-scale applications, with key players aggressively filing broad intellectual property portfolios to secure technological moats and future market positioning.
Core strategic imperatives include forming symbiotic partnerships between agile biotech startups and large pharmaceutical or industrial biotechnology companies. This collaboration provides essential capital, manufacturing scale, and regulatory expertise. Success hinges on platform modularity, enabling rapid customization for diverse applications. Companies are strategically focusing on high-value, low-volume therapeutics initially to prove efficacy and safety, planning subsequent expansion into broader, scalable industrial and environmental monitoring applications.
Analyst Key Takeaways:
Synthetic protein circuits represent a rapidly advancing niche within synthetic biology, driven by the shift from gene-level regulation to more precise, real-time control at the protein level. Their ability to enable dynamic cellular responses, post-translational modifications, and complex signaling behaviors positions them as a critical tool in next-generation therapeutic design, including cell and gene therapies. Increasing integration with technologies such as CRISPR, AI-driven protein design, and modular biological engineering frameworks is accelerating innovation and expanding their applicability across both healthcare and industrial biotechnology.
From a market dynamics perspective, synthetic protein circuits are expected to outpace broader gene circuit development due to their higher functional specificity and scalability in complex biological systems. Demand is being reinforced by growing investments in programmable biology, rising adoption in biomanufacturing, and the need for more controllable and adaptive biological systems. While still emerging, this segment is strategically important, with strong potential to become a foundational layer in advanced synthetic biology platforms as research transitions into commercial and clinical deployment.
REGIONAL TRENDS AND GROWTH
North America leads the market, fueled by substantial venture capital, strong academic hubs, and a robust biotechnology ecosystem. Europe follows, with significant initiatives in engineered cell therapies and ethical regulatory frameworks. The Asia-Pacific region is the fastest-growing, driven by heavy government investment in synthetic biology in China, Singapore, and Japan, aiming to build domestic biotech sovereignty and address regional healthcare and manufacturing needs through strategic national bio-economy plans.
Primary growth drivers are the unmet need for precise therapeutics and advancements in computational protein design. Key restraints involve high development costs, technical complexity, and evolving regulatory pathways for genetically engineered circuits. Opportunities abound in next-generation diagnostics and smart biomanufacturing. Major challenges include ensuring long-term circuit stability in vivo, managing potential immunogenicity, and scaling production consistently, all while navigating public perception and ethical considerations surrounding engineered biological systems.
SYNTHETIC PROTEIN CIRCUITS MARKET SEGMENTATION ANALYSIS
BY TYPE:
The type-based segmentation of the Synthetic Protein Circuits Market plays a foundational role in defining how solutions are designed, optimized, and commercialized. Enzyme-based circuits, transcription factor circuits, signal transduction circuits, feedback control circuits, oscillatory protein circuits, and modular protein circuits represent the major structural formats used across industries. The dominant factor in this segment is functional precision, as different circuit types are engineered to perform specific biological logic operations with high accuracy. Enzyme-based and transcription factor circuits often dominate due to their reliability in gene regulation and metabolic pathway control, while modular circuits gain traction because of their scalability and reusability across multiple biological environments.
Another critical growth driver within this segment is technological adaptability and integration capability. Oscillatory and feedback control circuits are increasingly preferred in advanced therapeutic and biosensing applications because they provide dynamic control over protein expression. The rise of programmable biology and synthetic gene networks has significantly strengthened demand for modular and signal-based circuit types, enabling customization and faster innovation cycles. Market expansion is further influenced by research funding, cross-disciplinary collaboration, and the continuous need for more stable and predictable circuit architectures in both industrial and clinical settings.
BY APPLICATION:
Application segmentation highlights the diverse utility of synthetic protein circuits across medical therapeutics, drug discovery, industrial biotechnology, agricultural biotechnology, environmental monitoring, and biosensing & diagnostics. The dominant factor in this segment is healthcare and pharmaceutical demand, as therapeutic protein circuits enable targeted drug delivery, disease detection, and precision medicine strategies. Drug discovery and diagnostics applications lead market adoption because they directly reduce research timelines and improve molecular screening efficiency, which is crucial for pharmaceutical competitiveness.
A second major influencing factor is industrial and environmental sustainability needs. Industrial biotechnology applications benefit from protein circuits that optimize fermentation, enzyme production, and metabolic engineering processes, while environmental monitoring circuits are increasingly used to detect pollutants and toxins in real time. Agricultural biotechnology also shows strong growth due to rising food security concerns and the demand for genetically enhanced crops. The broad applicability of protein circuits across multiple verticals ensures steady market momentum and diversified revenue streams.
BY END USER:
The end-user segmentation includes pharmaceutical companies, biotechnology firms, academic & research institutes, contract research organizations, healthcare institutions, and government laboratories. The primary dominant factor here is research intensity and funding availability, with pharmaceutical and biotechnology companies leading due to their continuous investment in innovative biologics and synthetic biology platforms. Academic institutions and government laboratories also play a crucial role by generating foundational research and early-stage prototypes that later transition into commercial solutions.
Another strong driver is outsourcing and collaboration trends. Contract research organizations gain market relevance because they provide specialized expertise and cost-effective experimental infrastructure. Healthcare institutions contribute through clinical testing and diagnostic integration, creating a bridge between research and real-world implementation. The expanding ecosystem of partnerships among these end users accelerates technology transfer, reduces development cycles, and supports scalable adoption of synthetic protein circuits globally.
BY COMPONENT:
Component-based segmentation consists of protein sensors, regulatory elements, expression vectors, reporter proteins, switch modules, and scaffold proteins. The dominant factor in this segment is system functionality and accuracy, as each component contributes to the overall performance and responsiveness of the protein circuit. Protein sensors and regulatory elements hold a significant share due to their role in detecting biological signals and controlling gene expression, which are critical for circuit reliability and precision.
A secondary but equally important factor is component interoperability and modular design. Expression vectors and scaffold proteins enable structural stability and efficient protein assembly, while switch modules and reporter proteins enhance monitoring and decision-making capabilities within synthetic systems. The demand for standardized, reusable components continues to grow as laboratories and companies aim to reduce complexity, improve reproducibility, and speed up development cycles in synthetic biology applications.
BY TECHNOLOGY:
Technology segmentation includes CRISPR-based engineering, recombinant DNA technology, cell-free protein synthesis, directed evolution, computational protein design, and microfluidic integration. The most dominant factor is precision engineering capability, with CRISPR and computational design tools leading due to their ability to deliver targeted modifications and predictive modeling. These technologies significantly reduce experimental uncertainty and enable rapid prototyping of complex protein circuits.
Another major influence is automation and scalability potential. Cell-free synthesis and microfluidic integration allow high-throughput experimentation, which is essential for industrial and pharmaceutical scalability. Directed evolution remains critical for optimizing protein stability and performance under varying conditions. Continuous technological convergence with artificial intelligence and machine learning further strengthens innovation pipelines and accelerates commercialization.
BY CIRCUIT COMPLEXITY:
This segment includes single-layer circuits, multi-layer circuits, logic gate circuits, toggle switch circuits, memory circuits, and hybrid circuits. The dominant factor is functional sophistication, as industries increasingly demand circuits capable of performing multi-step biological computations. Multi-layer and logic gate circuits dominate advanced research environments because they allow complex signal processing and decision-making within biological systems.
A second growth driver is application-specific customization. Memory and toggle switch circuits are gaining traction in therapeutic and diagnostic applications where long-term stability and reversible control are essential. Hybrid circuits also show strong potential as they combine multiple functional layers into unified platforms. The rising complexity of biological challenges naturally fuels demand for more advanced circuit architectures.
BY DELIVERY METHOD:
Delivery method segmentation consists of viral vectors, non-viral vectors, lipid nanoparticles, electroporation, microinjection, and biolistic delivery. The primary dominant factor is delivery efficiency and safety profile, with viral and lipid-based systems leading due to their higher transfection success rates. Pharmaceutical and therapeutic applications heavily rely on safe and targeted delivery mechanisms to ensure regulatory compliance and clinical effectiveness.
Another critical factor is cost and scalability of deployment. Non-viral methods such as electroporation and microinjection are preferred in laboratory settings for their precision and lower biosafety risks. Biolistic delivery remains important in agricultural biotechnology due to its effectiveness in plant cell modification. Continuous innovation in nano-delivery systems and biomaterial compatibility significantly shapes the growth trajectory of this segment.
BY PROTEIN FUNCTION:
Protein function segmentation includes catalytic, binding, structural, transport, regulatory, and fluorescent/reporter proteins. The dominant factor here is biological role specialization, as different protein functions directly influence circuit outcomes. Catalytic and regulatory proteins hold the largest share because they actively drive biochemical reactions and gene expression control, which are central to circuit operations.
A secondary factor is monitoring and visualization capability. Fluorescent and reporter proteins are essential for real-time observation and validation of circuit performance, particularly in research and diagnostics. Binding and transport proteins also contribute to targeted molecule interaction and signal propagation. The functional diversity within this segment ensures wide-ranging applications and continuous innovation opportunities.
BY INDUSTRY VERTICAL:
Industry vertical segmentation covers healthcare & life sciences, agriculture, food & beverage, environmental science, chemical industry, and energy & biofuels. The dominant factor is healthcare sector expansion, where synthetic protein circuits enable next-generation therapeutics, diagnostics, and personalized medicine. Life sciences research institutions and pharmaceutical industries drive the highest revenue contribution due to consistent innovation demand.
Another influential factor is sustainability and efficiency requirements in non-medical sectors. Agriculture and food industries utilize protein circuits to enhance crop yield and nutritional value, while environmental and energy sectors employ them for waste reduction and biofuel optimization. The cross-industry adaptability of protein circuits ensures long-term market resilience and diversification.
RECENT DEVELOPMENTS
- In Jan 2024: SyntheX and Bristol Myers Squibb expanded their collaboration, aiming to discover novel oncology therapeutics using synthetic protein switch technology, signaling strong pharmaceutical validation.
- In May 2024: Gingko Bioworks acquired Circularis Biotechnologies to enhance its platform for discovering genetic parts and designing advanced, tunable synthetic gene circuits for cell engineering programs.
- In Aug 2024: Standigm, leveraging AI for circuit design, partnered with a major chemical company to optimize metabolic pathways for sustainable chemical production using synthetic protein-based logic gates.
- In Nov 2024: A research consortium led by ETH Zurich published a breakthrough in Nature demonstrating a synthetic protein circuit capable of real-time discrimination and elimination of specific cancer cell subtypes.
- In Feb 2025: Asimov announced a strategic partnership with a leading CDMO to scale the manufacturing of mammalian cells programmed with advanced protein circuits for cell therapy applications.
KEY PLAYERS ANALYSIS
- Ginkgo Bioworks
- SyntheX
- Asimov
- Senti Biosciences
- Sana Biotechnology
- Cellarity
- Synlogic
- Precisement
- Standigm
- Novartis AG
- Bristol Myers Squibb
- GlaxoSmithKline plc
- Merck KGaA
- Thermo Fisher Scientific Inc.
- Codexis, Inc.
- Arzeda
- Plexium
- Proteinea
- Serotiny
- PvP Biologics