According to insights from Real Time Data Stats, the Digital Molecular Neurodiagnostics Market was valued at USD 0.90 billion in 2025. It is expected to grow from USD 1.00 billion in 2026 to USD 2.4 billion by 2033, registering a CAGR of 13% during the forecast period (2026–2033).
MARKET SIZE AND SHARE
The digital molecular neurodiagnostics market is expanding rapidly, driven by the rising burden of neurological disorders and the growing use of AI-based analytics. The sector already holds multi-billion-dollar value and is set to record strong, sustained growth. Market share is expected to concentrate among leading companies that invest in advanced diagnostic platforms and biomarker innovation. This shift marks a clear move away from conventional testing toward precise, data-driven solutions. North America will lead early adoption, supported by strong research infrastructure and high healthcare spending, ensuring a dominant revenue contribution in the initial phase.
Growth will accelerate as healthcare systems adopt digital tools and prioritize early, non-invasive detection of diseases such as Alzheimer’s and Parkinson’s. Emerging economies will steadily gain traction as access to advanced diagnostics improves and costs decline. The competitive landscape will tighten, with major firms acquiring innovative startups to strengthen their technology base and expand capabilities. Over time, the market will scale significantly in value, highlighting its critical role in transforming neurology care and enabling more personalized, predictive treatment approaches worldwide.
INDUSTRY OVERVIEW AND STRATEGY
This industry integrates molecular biology, digital tools, and neuroimaging to diagnose central nervous system disorders with unprecedented precision. It leverages biomarkers, genomic sequencing, and artificial intelligence to analyze complex neurological data. The sector is inherently interdisciplinary, merging biotechnology, information technology, and clinical neurology. Its core aim is to enable earlier, more accurate diagnoses, moving beyond symptomatic assessment to underlying molecular mechanisms, thereby revolutionizing patient stratification and treatment monitoring in cognitive and movement disorders.
Strategic focus centers on collaboration between diagnostic firms, tech giants, and research institutes to accelerate platform development. Key strategies include heavy R&D investment for novel biomarker panels, securing regulatory approvals for new assays, and forming partnerships with healthcare providers for clinical integration. Companies are also prioritizing data monetization strategies and developing scalable, cloud-based analysis platforms to secure a sustainable competitive advantage in this high-value diagnostic frontier.
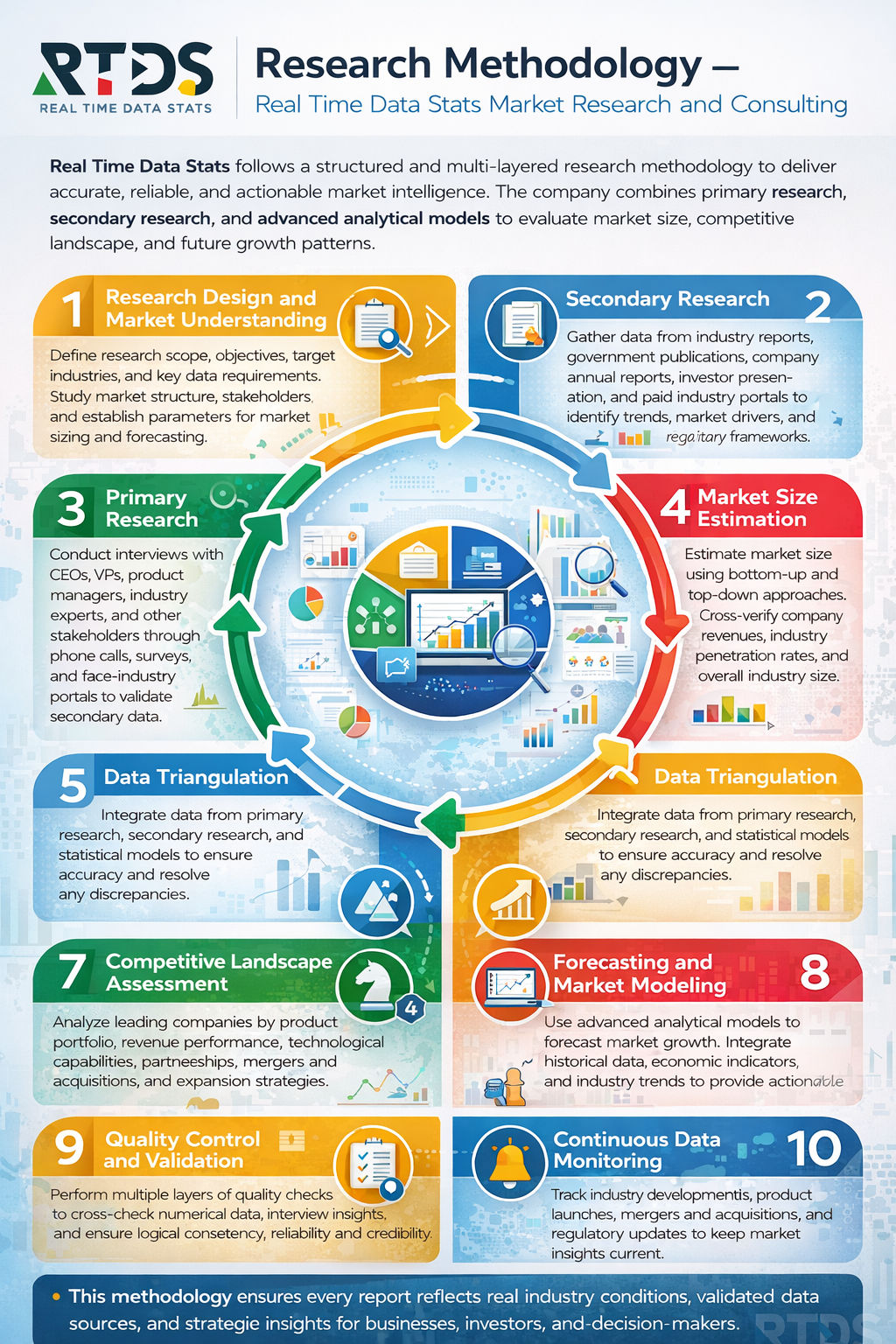
Analyst Key Takeaways:
The Digital Molecular Neurodiagnostics segment is emerging as a high-impact convergence layer within the broader neurodiagnostics ecosystem, driven by the integration of molecular biomarkers with AI-enabled diagnostic platforms. Adoption is being accelerated by the clinical shift toward early detection of neurodegenerative diseases such as Alzheimer’s and Parkinson’s, where traditional imaging and electrophysiology tools have limitations in sensitivity and timing. The increasing validation of blood-based biomarkers and next-generation sequencing techniques is reshaping diagnostic pathways, enabling less invasive, more scalable testing approaches.
From a growth dynamics perspective, the market is benefiting from disproportionately strong momentum compared to its parent segments due to digital augmentation and precision medicine trends. However, penetration remains structurally constrained by regulatory complexity, clinical validation requirements, and integration challenges across healthcare IT systems. Strategic investments are concentrated in biomarker discovery, AI-driven analytics, and companion diagnostics, positioning this niche as a critical enabler of next-generation neurology care rather than a standalone diagnostic category.
REGIONAL TRENDS AND GROWTH
North America commands the largest market share, propelled by advanced healthcare infrastructure, high R&D expenditure, and favorable reimbursement frameworks. Europe follows, with strong initiatives in genomic research and data governance. The Asia-Pacific region emerges as the fastest-growing market, driven by rising healthcare investment, increasing neurological disease burden, and improving diagnostic capabilities. Regional trends show a clear divergence in adoption speed, regulatory pathways, and partnership models between established and emerging economies.
Primary drivers include the aging global population, technological advancements in sequencing and AI, and growing precision medicine adoption. Significant restraints involve high diagnostic costs, data privacy concerns, and regulatory complexities. Opportunities lie in untapped emerging markets, portable point-of-care devices, and multi-omics integration. Key challenges encompass clinical validation of novel biomarkers, interoperability of digital health systems, and the need for skilled personnel to interpret complex molecular data.
DIGITAL MOLECULAR NEURODIAGNOSTICS MARKET SEGMENTATION ANALYSIS
BY TYPE:
Genomic diagnostics dominate the digital molecular neurodiagnostics market due to their strong ability to identify inherited neurological disorders and mutation-driven diseases. Growing adoption of whole-genome and targeted sequencing enables clinicians to detect disease susceptibility at an early stage. Increasing demand for precision neurology and declining sequencing costs further strengthen genomic diagnostics adoption across hospitals and specialized diagnostic centers, particularly for neurodegenerative and rare neurological conditions requiring molecular-level accuracy.
Transcriptomic, proteomic, epigenetic, biomarker-based, and multi-omics diagnostics expand rapidly as clinicians seek functional insights beyond DNA. These approaches allow deeper understanding of gene expression, protein interactions, and disease progression mechanisms. Rising investments in systems biology and data-driven neuroscience push adoption of multi-omics diagnostics, as they integrate multiple molecular layers to improve diagnostic confidence, therapy selection, and disease monitoring in complex neurological disorders.
BY TECHNOLOGY:
Next-generation sequencing and PCR-based technologies lead the market because they deliver high sensitivity, scalability, and reproducibility in neurological diagnostics. Continuous innovation in sequencing platforms enhances throughput and reduces turnaround time, making them suitable for both clinical and research environments. Digital PCR strengthens market growth by enabling absolute quantification of low-abundance biomarkers critical in early-stage neurological disease detection.
Microarray, CRISPR-based diagnostics, and lab-on-chip platforms gain traction due to their cost efficiency and portability. CRISPR diagnostics attract attention for rapid molecular detection and high specificity, while lab-on-chip solutions support decentralized testing. Growing demand for point-of-care neurodiagnostics and automation-driven workflows accelerates adoption of compact, high-precision technologies across emerging healthcare systems.
BY APPLICATION:
Neurodegenerative disease diagnostics represent the largest application segment, driven by the rising global burden of Alzheimer’s, Parkinson’s, and related disorders. Digital molecular tools support early detection, disease stratification, and progression tracking, which are essential for improving patient outcomes. Increasing clinical trial activity and demand for personalized therapeutic approaches further fuel adoption across neurology-focused healthcare facilities.
Neuro-oncology, neuroinfectious, neurodevelopmental, neurovascular, and psychiatric diagnostics expand steadily due to improved molecular understanding of brain disorders. Digital platforms enable accurate differentiation between overlapping neurological symptoms and underlying molecular causes. Growing awareness of molecular psychiatry and infection-driven neurological complications strengthens the role of advanced diagnostics in comprehensive neurological disease management.
BY DISEASE INDICATION:
Alzheimer’s disease and Parkinson’s disease drive substantial demand due to their high prevalence and diagnostic complexity. Digital molecular neurodiagnostics enable identification of disease-associated biomarkers, genetic mutations, and molecular signatures long before clinical symptoms appear. Early diagnosis becomes increasingly important as disease-modifying therapies advance, strengthening reliance on molecular diagnostics for patient stratification and treatment planning.
Multiple sclerosis, brain tumors, epilepsy, and amyotrophic lateral sclerosis benefit from molecular profiling that supports differential diagnosis and therapy optimization. Advanced diagnostics assist in identifying tumor genetics, inflammatory markers, and neurodegenerative pathways. Increasing integration of molecular diagnostics into clinical guidelines enhances adoption across specialized neurology and oncology centers worldwide.
BY END USER:
Hospitals and diagnostic laboratories dominate market adoption due to high testing volumes and access to advanced infrastructure. These settings integrate digital molecular platforms to improve diagnostic accuracy, reduce misdiagnosis, and support personalized treatment decisions. Increasing investments in hospital-based molecular laboratories and automation strengthen this segment’s leadership across developed and emerging healthcare markets.
Research institutes, academic medical centers, biotechnology companies, and pharmaceutical companies drive innovation-focused demand. These end users rely on digital molecular neurodiagnostics for biomarker discovery, drug development, and clinical trial optimization. Strong collaboration between academia and industry accelerates technology validation and commercial adoption, reinforcing the market’s long-term growth trajectory.
BY SAMPLE TYPE:
Blood and cerebrospinal fluid samples hold dominant positions due to their clinical relevance in neurological diagnostics. Blood-based molecular testing supports minimally invasive screening, while cerebrospinal fluid remains critical for central nervous system biomarker detection. Advances in liquid biopsy technologies significantly enhance diagnostic sensitivity, supporting broader clinical acceptance and routine use.
Saliva, tissue biopsies, urine, and cell-free DNA/RNA samples gain importance as non-invasive and repeatable diagnostic options. Cell-free nucleic acid analysis supports real-time disease monitoring and therapy response assessment. Growing emphasis on patient comfort and longitudinal disease tracking accelerates adoption of alternative sample types across digital neurodiagnostic workflows.
BY PLATFORM:
Cloud-based platforms lead the market due to their scalability, real-time data access, and integration with AI-driven analytics. These platforms support collaborative diagnostics, centralized data storage, and rapid interpretation of complex molecular datasets. Increasing adoption of digital health infrastructure and remote diagnostics strengthens cloud-based platform deployment globally.
On-premise, integrated digital systems, AI-enabled platforms, bioinformatics software, and point-of-care platforms maintain strong demand in regulated and data-sensitive environments. AI-enabled platforms enhance diagnostic precision by identifying complex molecular patterns, while point-of-care platforms support faster clinical decisions. Platform diversification ensures flexibility across varied healthcare settings and regulatory landscapes.
BY WORKFLOW:
Sample preparation and molecular amplification remain critical workflow stages, as accuracy at these steps directly impacts diagnostic outcomes. Automation in sample handling reduces human error and improves reproducibility. Increasing adoption of standardized workflows supports consistent diagnostic performance across high-throughput clinical laboratories and research environments.
Sequencing, data acquisition, analysis, interpretation, and reporting increasingly rely on advanced software and AI algorithms. Data-driven interpretation accelerates diagnosis and supports clinical decision-making. Integration of clinical decision support systems into diagnostic workflows enhances efficiency, reduces reporting time, and improves treatment alignment in neurological care pathways.
BY COMPONENT:
Instruments and reagents dominate revenue generation due to recurring demand and continuous technological upgrades. High-performance instruments ensure accuracy and throughput, while specialized reagents enable disease-specific molecular testing. Growing test volumes and expansion of molecular diagnostic laboratories drive sustained demand across both components.
Software solutions, consumables, assay panels, and data management tools experience rapid growth as diagnostics become increasingly data-intensive. Advanced software enables interpretation of complex molecular data, while assay panels support disease-focused testing. Rising emphasis on data security, interoperability, and regulatory compliance strengthens adoption of comprehensive digital diagnostic ecosystems.
RECENT DEVELOPMENTS
- In Jan 2024: Quest Diagnostics launched the first clinically available digital blood test for early Alzheimer's disease detection, leveraging proprietary biomarker algorithms.
- In Apr 2024: Roche partnered with LumiraDx to co-develop and commercialize rapid, point-of-care digital molecular tests for neurological conditions, enhancing decentralized diagnostics.
- In Jul 2024: Biogen and Apple announced a multi-year research collaboration to develop digital biomarkers for cognitive decline using Apple Watch sensor data and AI analytics.
- In Nov 2024: Abbott received FDA breakthrough device designation for its novel, portable digital panel designed to detect multiple neurodegenerative disease biomarkers from a single blood sample.
- In Feb 2025: C₂N Diagnostics and GE HealthCare entered a strategic alliance to integrate C₂N's precise amyloid-beta blood testing into GE's AI-powered neuroimaging platforms for comprehensive diagnosis.
KEY PLAYERS ANALYSIS
- Roche Diagnostics
- Abbott Laboratories
- Quest Diagnostics
- Biogen
- C₂N Diagnostics
- LumiraDx
- Quanterix
- Simoa (a Bio-Techne brand)
- Illumina, Inc.
- Pacific Biosciences
- NeuroSense Therapeutics
- Neuron23
- Genentech (Roche)
- Eli Lilly and Company
- AstraZeneca
- Novartis AG
- Pfizer Inc.
- Mayo Clinic Laboratories
- Bio-Rad Laboratories
- PerkinElmer, Inc.