Autonomous Synthetic Biology Market Size, Share, Trends and Growth Analysis; By Type: Automated Gene Synthesis Platforms, AI-Driven DNA Design Systems, Autonomous Cell Engineering Systems By Technology: Machine Learning-Based Biological Modeling, Deep Learning-Enabled Protein Engineering By Component: Software Platforms, Hardware Systems By Application: Drug Discovery and Development, Industrial Biomanufacturing; By Workflow Stage: Design and Modeling, Build and Assembly, Test and Validation; By End User: Biotechnology Companies, Pharmaceutical Manufacturers, Research and Academic Institutes; By Deployment Mode; By Level of Autonomy; By Product Type: Synthetic DNA Constructs, Engineered Microorganisms; By Regions, and Industry Forecast, Global Report 2025-2033
Report ID: RTDS1555
Historical Range: 2020-2024
Forecast Period: 2025-2033
No. of Pages: 300+
Industry: Pharma and Healthcare
-
0:00
According to insights from Real Time Data Stats, the Autonomous Synthetic Biology Market was valued at USD 0.76 billion in 2025. It is expected to grow from USD 1.0 billion in 2026 to USD 5.5 billion by 2033, registering a CAGR of 28% during the forecast period (2026–2033).
MARKET SIZE AND SHARE
The autonomous synthetic biology market grows fast as firms integrate AI with automated genetic design and assembly. Advanced tools speed discovery, cut errors, and scale lab output. Market share will consolidate among pioneers offering integrated platforms, while biotechnology and pharmaceutical leaders capture large portions through strong pipelines and partnerships. North America will lead revenue due to deep R&D funding, skilled talent, and active alliances between academia and industry. Continuous investment in robotics, data platforms, and standardized workflows will further strengthen competitive positions.
Market share will depend on clear technology gaps in cloud biofoundries and proprietary AI models. Companies that deliver full-stack systems—from digital design to automated strain build and testing—will lead adoption. Startups will push novel methods and niche tools, while established industrial biotech firms expand share through acquisitions and in-house builds. Control of the end-to-end biodesign pipeline will define long-term advantage. Ongoing progress in data quality, model accuracy, and scalable manufacturing will unlock new revenue streams and reshape leadership.
INDUSTRY OVERVIEW AND STRATEGY
The autonomous synthetic biology industry integrates artificial intelligence, robotic automation, and machine learning to automate the design, build, and test cycles of biological systems. This paradigm shift aims to accelerate R&D timelines for therapeutics, sustainable materials, and specialty chemicals. The strategic focus is on developing closed-loop platforms that minimize human intervention, thereby enhancing scalability, reproducibility, and innovation speed across diverse applications, from drug discovery to agricultural biotechnology.
Core competitive strategies revolve around building proprietary databases of genetic parts and optimized workflows. Key players are pursuing vertical integration and forming ecosystem partnerships with cloud providers and hardware manufacturers. Strategic emphasis is also placed on securing intellectual property around AI-driven design tools and standardized biological modules, creating high barriers to entry and establishing long-term customer lock-in through superior platform efficiency and predictive modeling capabilities.
Analyst Key Takeaways:
The Autonomous Synthetic Biology market is emerging as a high-impact subsegment within synthetic biology, driven by the convergence of artificial intelligence, laboratory automation, and advanced bioengineering workflows. The transition from manual experimentation to self-driving laboratories is significantly accelerating the design–build–test–learn (DBTL) cycle, improving reproducibility and reducing time-to-discovery. Adoption is being led by pharmaceutical, biotechnology, and industrial biology players seeking efficiency gains, scalable innovation, and data-driven experimentation. As a result, automation is shifting from a supportive tool to a core operational backbone in next-generation biological research.
From a growth perspective, the segment is expected to outpace the broader synthetic biology market due to its strong alignment with digital transformation trends and increasing investment in AI-enabled research infrastructure. Expansion of biofoundries, rising demand for precision engineering of biological systems, and integration of cloud-based lab platforms are reinforcing long-term growth momentum. However, scalability challenges, high initial capital requirements, and interoperability issues across platforms remain key constraints. Despite these barriers, continuous advancements in machine learning models, robotics, and lab standardization are expected to unlock wider commercial adoption over the forecast period.
REGIONAL TRENDS AND GROWTH
Regionally, North America leads, fueled by strong venture capital, top-tier research institutions, and a dense concentration of tech and biotech firms. Europe follows, emphasizing ethical frameworks and sustainable applications through significant public funding initiatives. The Asia-Pacific region emerges as the fastest-growing market, with governments actively investing in bio-manufacturing infrastructure and national genomics programs to achieve strategic independence in bioproduction and pharmaceuticals.
Primary growth drivers include the urgent need for biomanufacturing agility, pharmaceutical innovation demands, and sustainability goals. Key restraints are high initial capital costs, a scarcity of interdisciplinary talent, and evolving regulatory uncertainty. Major opportunities lie in pandemic preparedness, climate-resilient agriculture, and personalized medicine. Critical challenges involve addressing public acceptance, establishing global bio-safety standards, and managing the cybersecurity risks inherent in digitized biological systems.
AUTONOMOUS SYNTHETIC BIOLOGY MARKET SEGMENTATION ANALYSIS
BY TYPE:
Automated gene synthesis platforms drive strong demand by reducing biological design timelines and minimizing human intervention. These systems accelerate DNA assembly with precision, supporting rapid prototyping across pharmaceuticals and industrial biotech. Their ability to autonomously iterate genetic sequences improves experimental throughput while lowering operational error rates. Growing investment in high-speed synthesis and falling sequencing costs further strengthen adoption, especially among biofoundries seeking scalable and repeatable biological engineering processes.
AI-driven DNA design systems dominate innovation by enabling predictive and adaptive biological construction. These platforms leverage algorithmic learning to optimize gene constructs before physical synthesis, reducing trial-and-error cycles. Autonomous cell engineering systems complement this by executing real-time genetic modifications with minimal supervision. Together, self-optimizing biofoundries and robotic laboratory automation platforms enhance reproducibility, ensure consistency across experiments, and support large-scale deployment of synthetic biology solutions across diverse industries.
BY TECHNOLOGY:
Machine learning–based biological modeling forms the technological backbone of autonomous synthetic biology by transforming biological complexity into computable frameworks. These models improve pathway prediction, metabolic optimization, and protein folding accuracy. Deep learning–enabled protein engineering further enhances enzyme efficiency by analyzing massive biological datasets. The ability to continuously learn from experimental outcomes strengthens autonomous decision-making and shortens development cycles, making these technologies essential for advanced bioengineering workflows.
CRISPR-Cas autonomous editing technologies significantly influence market growth by enabling precise, scalable genome editing without manual oversight. Digital twin biology platforms replicate biological systems virtually, allowing real-time simulation and optimization before physical execution. High-throughput computational genomics and cloud-integrated synthetic biology systems support distributed experimentation, enhance collaboration, and ensure scalable processing power. These technologies collectively increase reliability, speed, and economic viability of autonomous synthetic biology operations.
BY COMPONENT:
Software platforms represent the most critical component, orchestrating design, execution, and optimization across autonomous biological workflows. These platforms integrate AI, data analytics, and modeling tools to manage complex biological experiments efficiently. Their ability to centralize decision-making and automate experimental logic improves reproducibility and accelerates innovation. As biological data volumes grow, demand for intelligent software capable of autonomous learning and optimization continues to expand rapidly.
Hardware systems and robotic workstations support physical execution by enabling high-throughput experimentation with minimal human interaction. Biological reagents tailored for automation ensure consistency across cycles, while data analytics engines extract actionable insights from experimental outputs. Cloud infrastructure services enable scalable computation and remote access, allowing organizations to deploy autonomous synthetic biology systems globally without heavy upfront infrastructure investments.
BY APPLICATION:
Drug discovery and development remain a leading application due to the need for faster target identification and optimized therapeutic design. Autonomous systems reduce compound screening timelines by continuously refining biological candidates based on experimental feedback. This approach lowers R&D costs and improves success rates, especially in complex biologics and personalized medicine. Pharmaceutical firms increasingly rely on autonomous synthetic biology to maintain competitive innovation pipelines.
Industrial biomanufacturing and agricultural bioengineering benefit from autonomous optimization of microbial strains and metabolic pathways. Environmental bioremediation and bioenergy production leverage self-optimizing organisms to improve efficiency under dynamic conditions. Diagnostic and therapeutic design applications further expand adoption by enabling rapid customization and adaptive response mechanisms, making autonomous synthetic biology a versatile solution across multiple high-impact industries.
BY WORKFLOW STAGE:
Design and modeling stages dominate value creation by defining biological outcomes before physical execution. Autonomous platforms analyze biological datasets to generate optimized genetic blueprints, reducing uncertainty early in development. Build and assembly stages benefit from automation that ensures precision and scalability. These stages form the foundation for reliable downstream experimentation and directly influence overall system performance.
Test, validation, and learn phases drive continuous improvement through closed-loop feedback mechanisms. Autonomous systems evaluate outcomes, refine parameters, and adapt strategies without manual intervention. Scale-up production and quality control stages further benefit from consistent execution and regulatory compliance. Together, workflow automation reduces bottlenecks, enhances reproducibility, and supports industrial-scale deployment of synthetic biology solutions.
BY END USER:
Biotechnology and pharmaceutical companies lead adoption due to their need for rapid innovation and cost-efficient R&D processes. Autonomous systems enable these organizations to explore complex biological designs while maintaining scalability and precision. Research and academic institutes also contribute significantly by using autonomous platforms to accelerate experimentation and knowledge discovery across genomics and systems biology.
Contract research organizations and industrial manufacturers increasingly adopt autonomous synthetic biology to enhance service efficiency and production consistency. Government and defense laboratories leverage these systems for biosecurity, advanced materials, and strategic research initiatives. Diverse end-user participation strengthens ecosystem maturity and accelerates commercialization across multiple application domains.
BY DEPLOYMENT MODE:
On-premise systems appeal to organizations requiring full data control and regulatory compliance, particularly in pharmaceutical and defense sectors. These deployments support customization and secure experimentation but involve higher capital investment. Cloud-based platforms, however, drive rapid adoption by offering scalability, reduced infrastructure costs, and collaborative accessibility across geographically distributed teams.
Hybrid deployment models combine control with flexibility, enabling organizations to balance security and scalability. Edge-integrated bio systems and remote laboratory automation support real-time experimentation in decentralized environments. Distributed biofoundry networks further expand deployment potential by enabling global collaboration, accelerating innovation, and optimizing resource utilization across autonomous synthetic biology ecosystems.
BY LEVEL OF AUTONOMY:
Semi-autonomous and supervised autonomous systems dominate early adoption by maintaining human oversight while automating repetitive processes. These systems reduce labor intensity and improve efficiency without fully removing operator control. They serve as transitional solutions for organizations gradually integrating autonomy into biological workflows.
Fully autonomous and self-learning bio platforms represent the future growth engine by enabling independent decision-making and adaptive optimization. Adaptive decision-making systems continuously refine experimental strategies, while closed-loop autonomous operations ensure consistent improvement. Higher autonomy levels enhance scalability, reduce human error, and unlock the full economic potential of synthetic biology automation.
BY PRODUCT TYPE:
Synthetic DNA constructs and engineered microorganisms account for significant market demand due to their role in therapeutic, industrial, and agricultural applications. Autonomous systems improve the accuracy and scalability of these products by optimizing genetic designs in real time. Synthetic proteins and enzymes benefit from predictive modeling that enhances functionality and stability.
Custom genetic circuits and cell-free synthetic systems support rapid prototyping and flexible production models. Bio-based chemical outputs further expand product diversity by offering sustainable alternatives to petrochemical processes. Autonomous optimization ensures consistent quality and performance, strengthening product commercialization across multiple end-use industries.
RECENT DEVELOPMENTS
- In Jan 2024: Ginkgo Bioworks launched a new automated foundry service targeting the sustainable production of key fragrance and food ingredients, expanding its industrial biotechnology portfolio.
- In Apr 2024: Synthego and a major cloud provider announced a strategic partnership to integrate advanced machine learning models with cloud-based bio-design tools, enhancing research scalability.
- In Jul 2024: Zymergen was acquired by a larger life sciences tools corporation, aiming to integrate its automation and AI-driven strain engineering platform into a broader product suite.
- In Nov 2024: Recursion Pharmaceuticals introduced an automated, high-throughput phenomics platform specifically for synthetic biology applications in novel therapeutic discovery.
- In Feb 2025: Twist Bioscience unveiled its next-generation silicon-based DNA synthesis platform, significantly increasing the speed and fidelity of gene fragment production for automated workflows.
KEY PLAYERS ANALYSIS
- Ginkgo Bioworks
- Twist Bioscience
- Synthego
- Amyris
- Zymergen
- Recursion Pharmaceuticals
- Codexis
- Synthace (a Antha company)
- Berkeley Lights
- Benchling
- GenScript Biotech
- Thermo Fisher Scientific
- Agilent Technologies
- Danaher (Cytiva)
- Merck KGaA
- Evonetix
- Arzeda
- Culture Biosciences
- Inscripta
- Nautilus Biotechnology
Autonomous Synthetic Biology Market Segmentation Analysis
By Type:
- Automated Gene Synthesis Platforms
- AI-Driven DNA Design Systems
- Autonomous Cell Engineering Systems
- Self-Optimizing Biofoundries
- Closed-Loop Biological Control Systems
- Robotic Laboratory Automation Platforms
By Technology:
- Machine Learning-Based Biological Modeling
- Deep Learning-Enabled Protein Engineering
- CRISPR-Cas Autonomous Editing Technologies
- Digital Twin Biology Platforms
- High-Throughput Computational Genomics
- Cloud-Integrated Synthetic Biology Systems
By Component:
- Software Platforms
- Hardware Systems
- Biological Reagents
- Robotic Workstations
- Data Analytics Engines
- Cloud Infrastructure Services
By Application:
- Drug Discovery and Development
- Industrial Biomanufacturing
- Agricultural Bioengineering
- Environmental Bioremediation
- Bioenergy Production
- Diagnostic and Therapeutic Design
By Workflow Stage:
- Design and Modeling
- Build and Assembly
- Test and Validation
- Learn and Optimization
- Scale-Up Production
- Quality Control and Compliance
By End User:
- Biotechnology Companies
- Pharmaceutical Manufacturers
- Research and Academic Institutes
- Contract Research Organizations
- Industrial Manufacturers
- Government and Defense Laboratories
By Deployment Mode:
- On-Premise Systems
- Cloud-Based Platforms
- Hybrid Deployment Models
- Edge-Integrated Bio Systems
- Remote Laboratory Automation
- Distributed Biofoundry Networks
By Level of Autonomy:
- Semi-Autonomous Systems
- Supervised Autonomous Platforms
- Fully Autonomous Biological Systems
- Self-Learning Bio Platforms
- Adaptive Decision-Making Systems
- Closed-Loop Autonomous Operations
By Product Type:
- Synthetic DNA Constructs
- Engineered Microorganisms
- Synthetic Proteins and Enzymes
- Custom Genetic Circuits
- Cell-Free Synthetic Systems
- Bio-Based Chemical Outputs
By Geography:
- North America (USA, Canada, Mexico)
- Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, Australia, South Korea, India, Rest of Asia-Pacific)
- South America (Brazil, Argentina, Rest of South America)
- Middle East and Africa (GCC Countries, South Africa, Rest of MEA)
Autonomous Synthetic Biology Market: Table of Contents
Introduction
- Market Definition and Scope
- Evolution of Autonomous Synthetic Biology
- Core Objectives of Autonomous Biological Systems
- Industry Ecosystem Overview
- Value Chain Analysis
- Market Terminology and Assumptions
Executive Summary
- Global Market Snapshot
- Key Growth Highlights
- Strategic Market Insights
- Investment Opportunity Overview
- Technology Adoption Summary
Market Dynamics
- Growth Drivers
- Market Restraints
- Emerging Opportunities
- Technology Challenges
Regulatory Landscape
- Ethical and Safety Considerations
- Market Trends and Innovation Landscape
- AI Integration in Synthetic Biology
- Autonomous Biofoundry Advancements
Closed-Loop Experimentation Trends
- Digital Twin Biology Adoption
- Robotics and Lab Automation Evolution
- Cloud-Native Biology Platforms
Autonomous Synthetic Biology Market Segmentation
- By Type:
- Automated Gene Synthesis Platforms
- AI-Driven DNA Design Systems
- Autonomous Cell Engineering Systems
- Self-Optimizing Biofoundries
- Closed-Loop Biological Control Systems
- Robotic Laboratory Automation Platforms
- By Technology:
- Machine Learning-Based Biological Modeling
- Deep Learning-Enabled Protein Engineering
- CRISPR-Cas Autonomous Editing Technologies
- Digital Twin Biology Platforms
- High-Throughput Computational Genomics
- Cloud-Integrated Synthetic Biology Systems
- By Component:
- Software Platforms
- Hardware Systems
- Biological Reagents
- Robotic Workstations
- Data Analytics Engines
- Cloud Infrastructure Services
- By Application:
- Drug Discovery and Development
- Industrial Biomanufacturing
- Agricultural Bioengineering
- Environmental Bioremediation
- Bioenergy Production
- Diagnostic and Therapeutic Design
- By Workflow Stage:
- Design and Modeling
- Build and Assembly
- Test and Validation
- Learn and Optimization
- Scale-Up Production
- Quality Control and Compliance
- By End User:
- Biotechnology Companies
- Pharmaceutical Manufacturers
- Research and Academic Institutes
- Contract Research Organizations
- Industrial Manufacturers
- Government and Defense Laboratories
- By Deployment Mode:
- On-Premise Systems
- Cloud-Based Platforms
- Hybrid Deployment Models
- Edge-Integrated Bio Systems
- Remote Laboratory Automation
- Distributed Biofoundry Networks
- By Level of Autonomy:
- Semi-Autonomous Systems
- Supervised Autonomous Platforms
- Fully Autonomous Biological Systems
- Self-Learning Bio Platforms
- Adaptive Decision-Making Systems
- Closed-Loop Autonomous Operations
- By Product Type:
- Synthetic DNA Constructs
- Engineered Microorganisms
- Synthetic Proteins and Enzymes
- Custom Genetic Circuits
- Cell-Free Synthetic Systems
- Bio-Based Chemical Outputs
Competitive Landscape
- Market Share Analysis
- Competitive Benchmarking
- Strategic Partnerships
- Mergers and Acquisitions
- Product Innovation Strategies
- Company Profiling Framework
Regional Market Analysis
- North America Market Overview
- Europe Market Overview
- Asia Pacific Market Overview
- Latin America Market Overview
- Middle East Market Overview
- Africa Market Overview
Investment and Funding Analysis
- Venture Capital Trends
- Government Funding Initiatives
- Corporate Investment Patterns
- Startup Ecosystem Analysis
Future Outlook
- Market Forecast Overview
- Technology Roadmap
- Long-Term Growth Opportunities
- Strategic Recommendations
Conclusion
- Market summary
- Key findings
- Strategic insights
Appendix
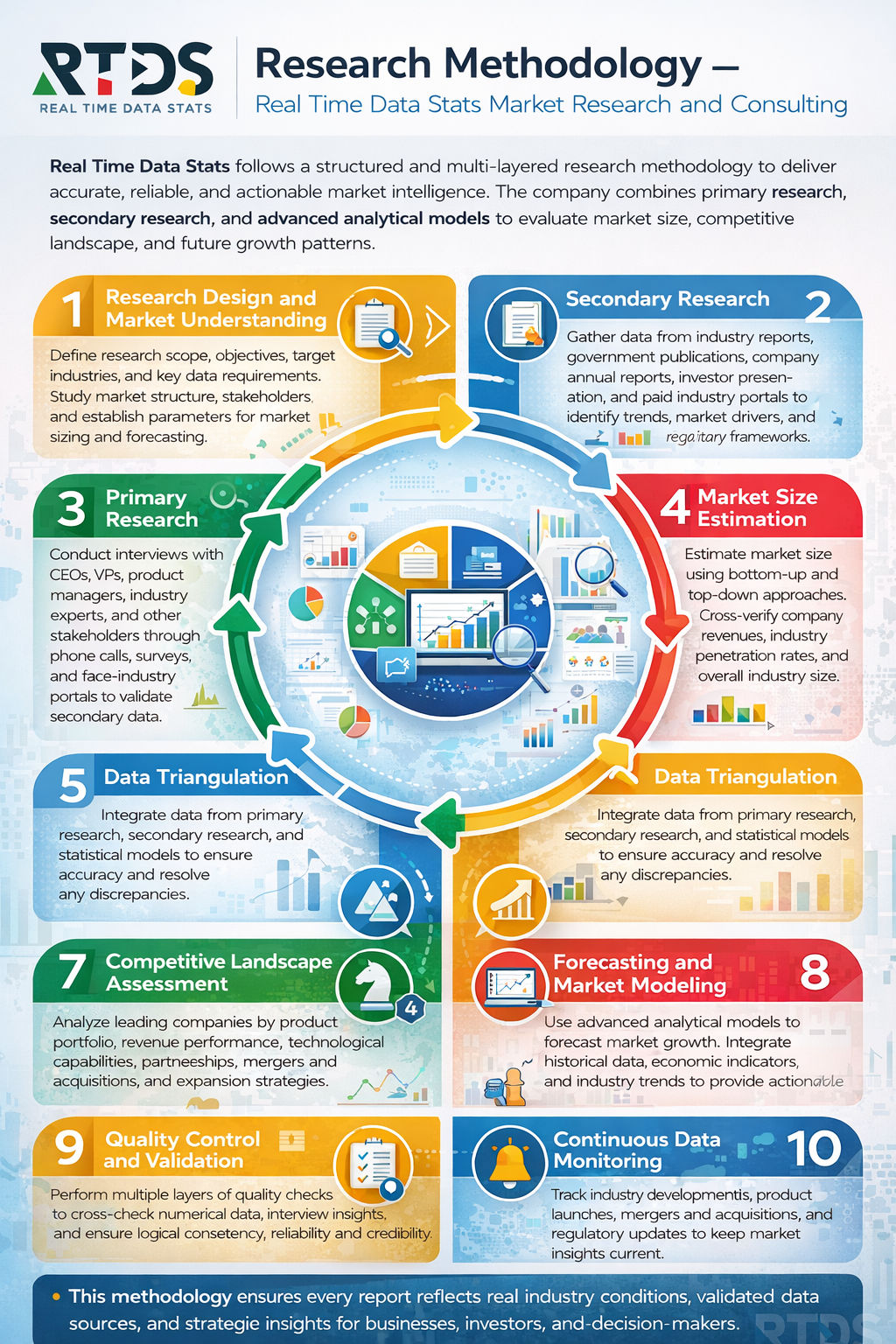
- Research Methodology
- Data Sources
- Abbreviations and Acronyms
- Disclaimer
List of Tables
- Table:1: Global Autonomous Synthetic Biology Market Overview
- Table:2: Market Dynamics Impact Analysis
- Table:3: Market Segmentation by Type
- Table:4: Market Segmentation by Technology
- Table:5: Market Segmentation by Component
- Table:6: Market Segmentation by Application
- Table:7: Market Segmentation by Workflow Stage
- Table:8: Market Segmentation by End User
- Table:9: Market Segmentation by Deployment Mode
- Table:10: Market Segmentation by Level of Autonomy
- Table:11: Market Segmentation by Product Type
- Table:12: Regional Market Share Analysis
- Table:13: Competitive Landscape Overview
- Table:14: Key Company Strategic Analysis
- Table:15: Market Forecast Summary
List of Figures
- Figure:1: Autonomous Synthetic Biology Market Ecosystem
- Figure:2: Market Value Chain Analysis
- Figure:3: Market Drivers and Restraints Framework
- Figure:4: Technology Adoption Curve
- Figure:5: Autonomous Biofoundry Workflow
- Figure:6: AI-Driven Synthetic Biology Architecture
- Figure:7: Market Segmentation Overview
- Figure:8: Regional Market Distribution
- Figure:9: Competitive Positioning Matrix
- Figure:10: Investment and Funding Landscape
- Figure:11: Autonomous System Maturity Levels
- Figure:12: Product Development Lifecycle
- Figure:13: Application Landscape Mapping
- Figure:14: Strategic Growth Opportunities
- Figure:15: Future Market Roadmap
Autonomous Synthetic Biology Market Key Factors
Drivers:
- Growing demand for sustainable biomaterials and bio-based production accelerates adoption.
- Major pharmaceutical and chemical companies increase investments in automated bio-foundries.
- Advancements in artificial intelligence and machine learning enhance design and optimization cycles.
Restraints:
- High capital investment for automated platforms limits access for small-scale researchers.
- Unclear regulatory frameworks and biosafety concerns delay commercial product pathways.
- A significant shortage of skilled personnel adept in both biology and engineering slows implementation.
Opportunities:
- Expanding applications in climate tech, like carbon capture and alternative fuels, open new markets.
- Cloud-based platforms and ""lab-as-a-service"" models democratize access for a broader user base.
- Convergence with nanotechnology and advanced analytics enables novel product development.
Challenges:
- Technical hurdles in standardizing biological parts and automating complex workflows persist.
- Ethical debates and public perception regarding genome editing and autonomous systems require careful navigation.
- Managing and securing vast amounts of sensitive genomic data presents a critical hurdle.
Autonomous Synthetic Biology Market Key Regional Trends
North America:
- Strong venture capital funding fuels startup growth and specialized foundry services.
- Defense and space agencies actively fund research for resilient biomanufacturing and in-situ resource utilization.
- Leading academic institutes establish public-private partnerships to set technical and ethical standards.
Europe:
- Stringent regulations push development of safe-by-design principles and traceability in automated workflows.
- Focus increases on circular economy applications, driving sustainable production of chemicals and textiles.
- Cross-border consortia form to build shared infrastructure and compete in the global bioeconomy.
Asia-Pacific:
- Governments implement national bio-strategies and invest heavily in industrial-scale synthetic biology infrastructure.
- Rapid growth in contract research and manufacturing services for global pharmaceutical and agrichemical firms.
- Significant research advances occur in microbial engineering for agriculture and waste valorization.
Frequently Asked Questions
Key Questions Answered
- What is the size and growth rate of the market industry?
- What are the main drivers influencing market growth trends?
- What are the key challenges and restraints faced by the market?
- What are the emerging trends and opportunities shaping the market?
- Who are the top players, and what strategies drive their success?
- What are the customer demographics and their buying behaviors?
- What are the key market segments, and what is their scope?
- What are the geographical dynamics, and which region dominates?
- What are the market forecasts and projections for the next decade?
- What regulatory factors are critical to consider for market growth?
Why Choose our Company?
- Major facts and stats backing every report we offer.
- Comprehensive analysis tailored to your business needs.
- Accurate insights backed by extensive market research.
- Expert support to address your queries promptly.
- Data-driven strategies for better decision-making.
- In-depth reports with actionable
recommendations. - Reliable statistics from trusted industry
sources. - Customizable reports to suit your specific goals.
- Unmatched quality and precision in market analysis.
- Competitive pricing with premium content delivery.
Share your experience!
We value you feedback. Please take a moment to review your experience with us.
Jump to Content
Company Services
Legal Help
We Accept
Business Contact
Call us
+917020482655
Need live support?
sales@realtimedatastats.com
Office address
IT Square, Hinjewadi, Pune - 511057 (Maharashtra- India)
Copyright ©2026 Real Time Data Stats. All rights reserved.