Autonomous Genomic Synthesis Market Size, Share, Trends and Growth Analysis; By Type: DNA Synthesizers, RNA Synthesizers, Oligonucleotide Synthesizers, Peptide Nucleic Acid Synthesizers; By Application: Drug Discovery, Vaccine Development, Synthetic Biology Research, Agricultural Biotechnology; By End User: Pharmaceutical Companies, Biotechnology Companies, Academic & Research Institutes, Contract Research Organizations; By Technology: Microarray-Based Synthesis, Enzymatic DNA Synthesis, Solid-Phase Synthesis, AI-Driven Genomic Design; By Component; By Workflow Stage; By Throughput; By Deployment Mode; By Industry Vertical; By Regions, and Industry Forecast, Global Report 2025-2033
Report ID: RTDS1519
Historical Range: 2020-2024
Forecast Period: 2025-2033
No. of Pages: 300+
Industry: Life Sciences & Biotechnology
-
0:00
According to insights from Real Time Data Stats, the Autonomous Genomic Synthesis Market was valued at USD 0.42 billion in 2025. It is expected to grow from USD 0.53 billion in 2026 to USD 2.45 billion by 2033, registering a CAGR of 24.4% during the forecast period (2026–2033).
MARKET SIZE AND SHARE
The autonomous genomic synthesis market is transitioning from a specialized niche technology to a mainstream tool within biotechnology and life sciences. Market growth is driven by declining synthesis costs, improved automation, and rising demand for synthetic biology applications in healthcare, agriculture, and industrial research. Early market share is concentrated among pioneering biotechnology firms and specialized synthetic biology companies that commercialized automated genome design and synthesis platforms. Increasing adoption in research institutions and commercial laboratories is further accelerating technological validation and scalability.
Over time, market share distribution is expected to shift as large pharmaceutical and chemical companies enter through acquisitions, partnerships, and internal platform development. While new entrants may increase competition, established players with integrated workflows, advanced automation systems, and extensive genomic libraries are likely to maintain strong positions. By the long-term forecast period, the market is projected to expand significantly, with North America and Europe holding substantial revenue shares due to strong R&D infrastructure, regulatory support, and sustained investment in advanced biotechnologies.
INDUSTRY OVERVIEW AND STRATEGY
The autonomous genomic synthesis industry represents the convergence of robotics, artificial intelligence, and molecular biology to automate the design-build-test-learn cycle for genetic material. It eliminates manual bottlenecks, enabling rapid prototyping of genes, pathways, and organisms for applications in therapeutics, agriculture, and industrial bios manufacturing. The core value proposition is unprecedented speed, scale, and reproducibility in genetic engineering, fundamentally accelerating research and development timelines across the life sciences sector.
Key strategic focuses for industry players include vertical integration, encompassing software for AI-driven DNA design, proprietary synthesis hardware, and downstream validation services. Partnerships with pharmaceutical giants and agrochemical firms are crucial for co-development and market penetration. Intellectual property strategy around enzymatic synthesis methods and machine learning algorithms forms a critical competitive moat. Companies are also strategically investing in cloud-platform connectivity to offer synthesis-as-a-service, democratizing access for smaller research entities and fueling ecosystem growth.
Analyst Key Takeaways:
The Autonomous Genomic Synthesis market is witnessing strong momentum due to the rapid convergence of artificial intelligence, robotic laboratory automation, and synthetic biology platforms. Increasing demand for high-throughput DNA construction, automated genome assembly, and precision gene engineering is accelerating adoption across pharmaceutical research, industrial biotechnology, agricultural genomics, and advanced therapeutics development. Companies are increasingly investing in autonomous biofoundries and closed-loop synthesis systems to reduce turnaround time, improve sequence accuracy, and enable scalable genomic engineering workflows.
North America currently leads innovation owing to strong biotechnology infrastructure, growing bioinformatics capabilities, and rising investments in AI-driven life science research. Meanwhile, Asia-Pacific is emerging as a high-growth region supported by expanding genomics research initiatives and increasing government funding for synthetic biology innovation. Strategic collaborations between biotech firms, cloud-AI providers, and laboratory automation companies are expected to intensify competitive dynamics, while advancements in enzymatic DNA synthesis and machine-learning-guided sequence optimization continue to reshape next-generation genomic manufacturing capabilities.
REGIONAL TRENDS AND GROWTH
Regionally, North America leads, propelled by substantial venture capital, a robust biotech sector, and supportive regulatory frameworks for synthetic biology. Europe follows closely, with strong academic-industry collaborations and initiatives like the European Union’s circular bioeconomy focus driving adoption. The Asia-Pacific region is poised for the highest growth rate, fueled by significant government investments in biomanufacturing, particularly in China and Singapore, aiming for technological sovereignty and industrial biotechnology leadership.
Primary growth drivers include the escalating demand for personalized medicine and mRNA-based therapeutics, which require rapid, accurate DNA synthesis. Key restraints are high initial capital expenditure and evolving biosecurity regulations governing synthetic DNA. Significant opportunities lie in scalable bio-production of novel materials, sustainable fuels, and climate-resilient crops. However, the industry faces challenges in standardizing protocols, managing ethical concerns around genome editing, and navigating complex, fragmented international biosafety and trade regulations that could hinder global market cohesion.
AUTONOMOUS GENOMIC SYNTHESIS MARKET SEGMENTATION ANALYSIS
BY TYPE:
The type-based segmentation of the Autonomous Genomic Synthesis Market reflects the growing diversification of synthesis needs across biological complexity levels. DNA synthesizers dominate due to their foundational role in gene construction, pathway engineering, and diagnostic assay development. RNA synthesizers are rapidly gaining traction, driven by expanding mRNA vaccine pipelines, RNA therapeutics, and functional genomics research. Oligonucleotide synthesizers remain critical for primers, probes, and short genetic constructs, especially in high-throughput laboratories where speed and accuracy determine productivity. Meanwhile, peptide nucleic acid synthesizers serve niche but high-value applications in molecular diagnostics and antisense research, where superior binding stability and specificity offer clear performance advantages.
Automated gene assembly platforms and CRISPR-based synthesis systems represent the most transformative segment within this category. These systems integrate synthesis, assembly, and editing into unified workflows, significantly reducing manual intervention and cycle times. CRISPR-based synthesis systems, in particular, benefit from rising investments in genome editing, functional genomics, and therapeutic target validation. The dominant growth factor across all types is the push toward fully autonomous, error-minimized, and scalable synthesis, allowing organizations to shift from experimental iteration to industrial-grade genetic manufacturing.
BY APPLICATION:
Application-based segmentation highlights the expanding scope of autonomous genomic synthesis beyond traditional research settings. Drug discovery leads adoption, as pharmaceutical companies rely on rapid gene synthesis to accelerate target validation, protein engineering, and lead optimization. Vaccine development follows closely, supported by global preparedness initiatives and the proven success of synthetic biology–driven vaccine platforms. Synthetic biology research remains a core application area, leveraging automated synthesis to design metabolic pathways, synthetic circuits, and novel biological systems with unprecedented speed.
Emerging applications such as agricultural biotechnology, personalized medicine, and industrial biotechnology are reshaping market demand. In agriculture, autonomous synthesis enables rapid trait engineering, stress resistance development, and microbial crop enhancers. Personalized medicine benefits from on-demand gene synthesis tailored to patient-specific genetic profiles, particularly in cell and gene therapies. Industrial biotechnology increasingly uses synthesized genes to optimize enzymes for bio-based chemicals, materials, and fermentation processes. The dominant driver across applications is the need for faster innovation cycles combined with reproducible, high-fidelity genetic outputs.
BY END USER:
End-user segmentation reflects varying operational scales and innovation objectives. Pharmaceutical and biotechnology companies dominate market share due to their capital-intensive R&D pipelines and strong demand for automated, compliant, and scalable synthesis solutions. These organizations prioritize platforms that reduce development timelines while ensuring regulatory traceability. Academic and research institutes represent a significant adoption base, driven by funding for genomics, systems biology, and translational research, where flexibility and multi-project support are critical.
Contract research organizations and clinical laboratories are emerging as high-growth end users, fueled by outsourcing trends and the rise of precision diagnostics. CROs benefit from autonomous systems that improve throughput and reduce labor dependency, enabling them to serve multiple clients efficiently. Government research agencies contribute steady demand through national genomics initiatives, biodefense programs, and public health research. The dominant factor influencing end-user adoption is the need to balance cost efficiency with advanced automation and data integrity.
BY TECHNOLOGY:
Technology-based segmentation reveals a strong shift toward next-generation synthesis methodologies. Solid-phase synthesis remains widely used due to its maturity and reliability, particularly for oligonucleotide production. However, enzymatic DNA synthesis is gaining momentum as it addresses scalability and environmental concerns associated with chemical synthesis. Microarray-based synthesis supports parallel production of large oligo libraries, making it essential for high-throughput genomic screening and variant analysis.
Advanced technologies such as AI-driven genomic design, cloud-integrated automation platforms, and robotic liquid handling systems are redefining competitive differentiation. AI-driven tools optimize sequence design, minimize synthesis errors, and predict functional outcomes, significantly enhancing success rates. Cloud integration enables remote control, collaboration, and data analytics across global teams, while robotic systems ensure precision and reproducibility. The dominant technological driver is the convergence of automation, intelligence, and connectivity into seamless genomic manufacturing ecosystems.
BY COMPONENT:
Component segmentation underscores the interdependence of physical infrastructure and digital intelligence. Hardware systems form the backbone of autonomous synthesis, encompassing synthesizers, robotics, and integrated lab equipment. Reagents and consumables generate recurring revenue and remain critical for synthesis fidelity, consistency, and throughput. Microfluidic chips are increasingly important, as they enable miniaturized, high-speed reactions while reducing reagent consumption and operational costs.
Software and AI platforms represent the fastest-growing component segment, driven by the need for workflow orchestration, predictive analytics, and error correction. Automation controllers coordinate multi-step processes, ensuring synchronization across devices. Data storage and management tools address growing requirements for sequence traceability, regulatory compliance, and intellectual property protection. The dominant factor across components is the shift toward modular, upgradeable systems that support continuous innovation without full infrastructure replacement.
BY WORKFLOW STAGE:
Workflow-stage segmentation highlights where automation delivers the greatest value. Sequence design and oligo production are critical entry points, as errors at these stages propagate downstream costs. Autonomous platforms increasingly integrate AI-based design tools to optimize codon usage, minimize secondary structures, and enhance expression efficiency. Gene assembly and error correction stages benefit significantly from automation, reducing failure rates and manual troubleshooting.
Later stages such as cloning, amplification, validation, and quality control are gaining attention as regulatory scrutiny increases, especially in clinical and industrial applications. Automated validation ensures consistency, traceability, and compliance with quality standards. The dominant driver across workflow stages is the need for end-to-end automation that eliminates handoffs and maintains data continuity throughout the synthesis lifecycle.
BY THROUGHPUT:
Throughput-based segmentation reflects diverse operational needs. Low- and medium-throughput systems cater to academic labs and early-stage research environments where flexibility outweighs volume. High-throughput and ultra-high-throughput platforms dominate industrial settings, enabling rapid synthesis of thousands of sequences for screening, optimization, and large-scale production.
Parallel synthesis systems and on-demand synthesis units add strategic flexibility, allowing organizations to scale output dynamically based on project needs. On-demand units are particularly attractive for decentralized labs and time-sensitive applications. The dominant factor shaping this segment is the demand for scalable throughput without compromising accuracy, turnaround time, or cost efficiency.
BY DEPLOYMENT MODE:
Deployment mode segmentation reflects evolving infrastructure strategies. On-premises systems remain preferred by organizations handling sensitive data, proprietary research, or regulated workflows. However, cloud-connected and hybrid platforms are rapidly gaining adoption, enabling remote monitoring, collaborative design, and predictive maintenance. These models reduce upfront capital expenditure while improving system utilization.
Standalone units serve smaller labs with limited automation needs, whereas integrated lab automation suites appeal to large-scale facilities seeking seamless workflow integration. Remote-operated systems support global research operations and centralized control models. The dominant deployment driver is the balance between data security, operational flexibility, and cost optimization.
BY INDUSTRY VERTICAL:
Industry vertical segmentation demonstrates the broadening impact of autonomous genomic synthesis. Healthcare and life sciences dominate adoption due to drug development, diagnostics, and gene therapy applications. Agriculture leverages synthetic genomics for crop resilience, yield improvement, and microbial solutions. Environmental science applications focus on engineered organisms for bioremediation and sustainability initiatives.
Energy and biofuels, food and beverage biotechnology, and defense and biosecurity represent high-growth verticals. These sectors rely on autonomous synthesis to develop enzymes, bio-based materials, and rapid-response genetic solutions. The dominant factor across verticals is the ability of autonomous genomic synthesis to translate biological complexity into predictable, industrial-scale innovation.
RECENT DEVELOPMENTS
- In Jan 2024: Twist Bioscience launched its Express Genes service, significantly accelerating high-quality gene synthesis delivery to just 5-7 business days, enhancing R&D speed for therapeutic and industrial applications.
- In Mar 2024: DNA Script announced a strategic partnership with Nuclera to integrate enzymatic synthesis with digital protein expression, streamlining the workflow from DNA design to functional protein validation.
- In Jun 2024: Codex DNA introduced its BioXp NGS library preparation kit, automating and consolidating next-generation sequencing library prep directly from synthetic DNA on its BioXp system.
- In Nov 2024: Ansa Biotechnologies achieved a milestone by enzymatically synthesizing the first-ever 1500-base pair gene, demonstrating the commercial viability and scalability of its enzyme-based synthesis technology.
- In Feb 2025: Ginkgo Bioworks acquired a specialized AI-driven DNA design software firm, enhancing its Codebase and Foundry capabilities for autonomous design and optimization of synthetic genetic pathways.
KEY PLAYERS ANALYSIS
- Twist Bioscience
- Ginkgo Bioworks
- Codex DNA (a subsidiary of Danaher)
- DNA Script
- Ansa Biotechnologies
- GenScript Biotech
- Thermo Fisher Scientific
- Agilent Technologies
- Synthego
- Evonetix
- Nuclera
- Camena Bioscience
- Molecular Assemblies
- TeselaGen Biotechnology
- Cytiva
- Roche (Sequencing Solutions)
- ATUM
- Eurofins Genomics
- BioBasic
- OriCiro Genomics
Autonomous Genomic Synthesis Market Segmentation Analysis
By Type:
- DNA Synthesizers
- RNA Synthesizers
- Oligonucleotide Synthesizers
- Peptide Nucleic Acid Synthesizers
- Automated Gene Assembly Platforms
- CRISPR-Based Synthesis Systems
By Application:
- Drug Discovery
- Vaccine Development
- Synthetic Biology Research
- Agricultural Biotechnology
- Personalized Medicine
- Industrial Biotechnology
By End User:
- Pharmaceutical Companies
- Biotechnology Companies
- Academic & Research Institutes
- Contract Research Organizations
- Clinical Laboratories
- Government Research Agencies
By Technology:
- Microarray-Based Synthesis
- Enzymatic DNA Synthesis
- Solid-Phase Synthesis
- AI-Driven Genomic Design
- Cloud-Integrated Automation Platforms
- Robotic Liquid Handling Systems
By Component:
- Hardware Systems
- Software & AI Platforms
- Reagents & Consumables
- Microfluidic Chips
- Automation Controllers
- Data Storage & Management Tools
By Workflow Stage:
- Sequence Design
- Oligo Production
- Gene Assembly
- Error Correction
- Cloning & Amplification
- Validation & Quality Control
By Throughput:
- Low-Throughput Systems
- Medium-Throughput Systems
- High-Throughput Systems
- Ultra-High-Throughput Platforms
- Parallel Synthesis Systems
- On-Demand Synthesis Units
By Deployment Mode:
- On-Premises Systems
- Cloud-Connected Systems
- Hybrid Platforms
- Standalone Units
- Integrated Lab Automation Suites
- Remote-Operated Systems
By Industry Vertical:
- Healthcare & Life Sciences
- Agriculture
- Environmental Science
- Energy & Biofuels
- Food & Beverage Biotechnology
- Defense & Biosecurity
By Geography:
- North America (USA, Canada, Mexico)
- Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, Australia, South Korea, India, Rest of Asia-Pacific)
- South America (Brazil, Argentina, Rest of South America)
- Middle East and Africa (GCC Countries, South Africa, Rest of MEA)
Autonomous Genomic Synthesis Market: Table of Contents
Executive Summary
- Market Snapshot
- Key Findings
- Analyst Insights
- Winning Strategies
- Future Outlook
Market Introduction
- Market Definition
- Market Scope
- Market Objectives
- Stakeholders Overview
- Study Timeline
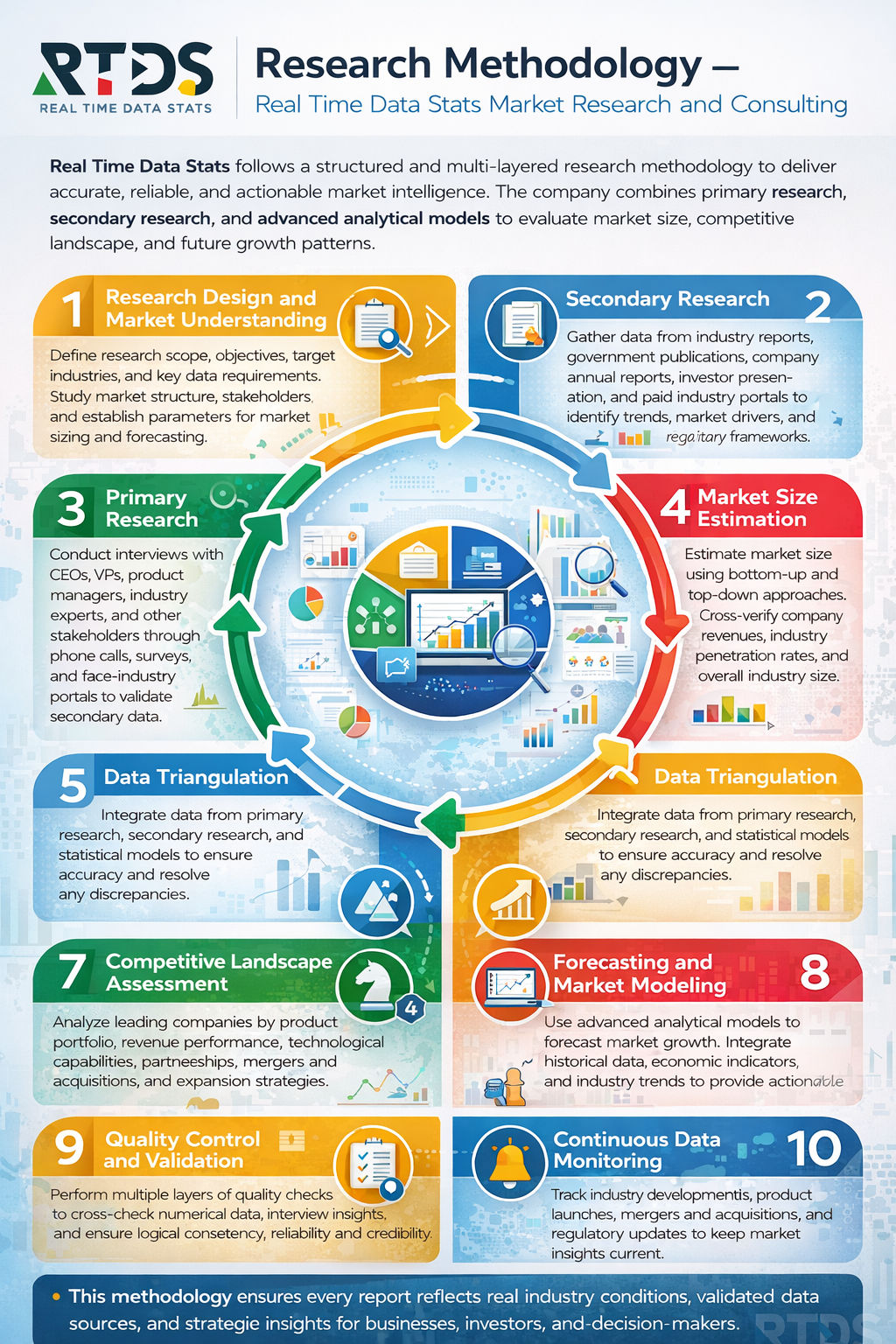
Research Methodology
- Data Collection Methods
- Primary Research
- Secondary Research
- Data Validation
- Assumptions & Limitations
Market Dynamics
- Drivers
- Restraints
- Opportunities
- Challenges
- Emerging Trends
Industry Analysis
- Value Chain Analysis
- Supply Chain Analysis
- Pricing Analysis
Regulatory Landscape
- Patent Analysis
- Strategic Analysis
- Porter’s Five Forces
- PESTLE Analysis
- Investment Analysis
- Risk Assessment
- Innovation Landscape
Autonomous Genomic Synthesis Market Segmentation Analysis
- By Type
- DNA Synthesizers
- RNA Synthesizers
- Oligonucleotide Synthesizers
- Peptide Nucleic Acid Synthesizers
- Automated Gene Assembly Platforms
- CRISPR-Based Synthesis Systems
- By Application
- Drug Discovery
- Vaccine Development
- Synthetic Biology Research
- Agricultural Biotechnology
- Personalized Medicine
- Industrial Biotechnology
- By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Academic & Research Institutes
- Contract Research Organizations
- Clinical Laboratories
- Government Research Agencies
- By Technology
- Microarray-Based Synthesis
- Enzymatic DNA Synthesis
- Solid-Phase Synthesis
- AI-Driven Genomic Design
- Cloud-Integrated Automation Platforms
- Robotic Liquid Handling Systems
- By Component
- Hardware Systems
- Software & AI Platforms
- Reagents & Consumables
- Microfluidic Chips
- Automation Controllers
- Data Storage & Management Tools
- By Workflow Stage
- Sequence Design
- Oligo Production
- Gene Assembly
- Error Correction
- Cloning & Amplification
- Validation & Quality Control
- By Throughput
- Low-Throughput Systems
- Medium-Throughput Systems
- High-Throughput Systems
- Ultra-High-Throughput Platforms
- Parallel Synthesis Systems
- On-Demand Synthesis Units
- By Deployment Mode
- On-Premises Systems
- Cloud-Connected Systems
- Hybrid Platforms
- Standalone Units
- Integrated Lab Automation Suites
- Remote-Operated Systems
- By Industry Vertical
- Healthcare & Life Sciences
- Agriculture
- Environmental Science
- Energy & Biofuels
- Food & Beverage Biotechnology
- Defense & Biosecurity
Competitive Landscape
- Market Share Analysis
- Company Benchmarking
- Strategic Developments
- Mergers & Acquisitions
- Partnerships & Collaborations
Company Profiles
- Company Overview
- Product Portfolio
- Financial Highlights
- Recent Developments
- SWOT Analysis
Forecast & Future Outlook
- Global Forecast
- Segment Forecast
- Regional Forecast
- Technology Forecast
- Investment Outlook
Conclusion
- Summary of Market Trends
- Key Drivers and Restraints
- Growth Opportunities
- Technology & Innovation Outlook
- Future Market Forecast
Appendix
- Abbreviations
- Data Sources
- Assumptions
- Disclaimer
List of Tables
- Table:1: Global Market Size Overview
- Table:2: Market Growth Rate Comparison
- Table:3: Revenue by Type
- Table:4: Revenue by Application
- Table:5: Revenue by End User
- Table:6: Revenue by Technology
- Table:7: Revenue by Component
- Table:8: Revenue by Workflow Stage
- Table:9: Revenue by Throughput
- Table:10: Revenue by Deployment Mode
- Table:11: Revenue by Industry Vertical
- Table:12: Regional Revenue Distribution
- Table:13: Competitive Market Share
- Table:14: Investment & Funding Analysis
- Table:15: Forecast Summary
List of Figures
- Figure:1: Market Research Framework
- Figure:2: Market Dynamics Overview
- Figure:3: Value Chain Diagram
- Figure:4: Supply Chain Flow
- Figure:5: Technology Evolution Map
- Figure:6: Segmentation by Type Chart
- Figure:7: Segmentation by Application Chart
- Figure:8: Segmentation by End User Chart
- Figure:9: Segmentation by Technology Chart
- Figure:10: Regional Market Share Map
- Figure:11: Competitive Positioning Matrix
- Figure:12: Investment Trend Graph
- Figure:13: Forecast Growth Curve
- Figure:14: Innovation Pipeline Illustration
- Figure:15: Future Opportunity Heatmap
Autonomous Genomic Synthesis Market Key Factors
Drivers:
- Increasing demand for rapid vaccine and therapeutic development accelerates adoption.
- Expanding synthetic biology applications in pharmaceuticals and industrial biotechnology push market growth.
- Advancements in AI and robotics significantly enhance system precision and throughput.
Restraints:
- High capital investment for automated platforms limits access for small research entities.
- Unclear regulatory frameworks for synthetically engineered organisms create adoption barriers.
- A shortage of skilled professionals proficient in both biology and engineering slows implementation.
Opportunities:
- Emerging bio-economy initiatives in various governments open new funding and application avenues.
- Potential to decentralize production for on-demand biologics and personalized medicines creates new models.
- Integration with cloud-based digital biology platforms expands access and collaborative potential.
Challenges:
- Addressing ethical and biosecurity concerns surrounding autonomous genetic design remains critical.
- Managing the complexity of integrating hardware, software, and biochemical processes poses technical hurdles.
- Ensuring data integrity and standardization across different platforms and workflows requires industry-wide effort.
Autonomous Genomic Synthesis Market Key Regional Trends
North America:
- Strong venture capital funding aggressively fuels startup innovation and scale-up.
- Leading pharmaceutical companies actively form partnerships with synthesis technology firms.
- Regulatory agencies are engaging earlier to shape pathways for lab-to-market translation.
Europe:
- Public research initiatives heavily emphasize ethical AI and responsible innovation in bio-automation.
- Focus is increasing on developing circular bioeconomy applications using sustainable feedstocks.
- Cross-border consortia are forming to build shared infrastructure and data repositories.
Asia-Pacific:
- Governments are making strategic investments to build domestic capacity and reduce import dependency.
- Contract research and manufacturing organizations are rapidly adopting automation to boost service offerings.
- Research is strongly targeting applications in agriculture and aquaculture to address food security.
Rest of the World:
- Key nations are exploring leapfrog opportunities in public health applications like local vaccine production.
- International partnerships are crucial for building initial foundational infrastructure and expertise.
- Focus remains on overcoming fundamental barriers like cost, connectivity, and cold-chain logistics.
Frequently Asked Questions
Key Questions Answered
- What is the size and growth rate of the market industry?
- What are the main drivers influencing market growth trends?
- What are the key challenges and restraints faced by the market?
- What are the emerging trends and opportunities shaping the market?
- Who are the top players, and what strategies drive their success?
- What are the customer demographics and their buying behaviors?
- What are the key market segments, and what is their scope?
- What are the geographical dynamics, and which region dominates?
- What are the market forecasts and projections for the next decade?
- What regulatory factors are critical to consider for market growth?
Why Choose our Company?
- Major facts and stats backing every report we offer.
- Comprehensive analysis tailored to your business needs.
- Accurate insights backed by extensive market research.
- Expert support to address your queries promptly.
- Data-driven strategies for better decision-making.
- In-depth reports with actionable
recommendations. - Reliable statistics from trusted industry
sources. - Customizable reports to suit your specific goals.
- Unmatched quality and precision in market analysis.
- Competitive pricing with premium content delivery.
Share your experience!
We value you feedback. Please take a moment to review your experience with us.
Jump to Content
Company Services
Legal Help
We Accept
Business Contact
Call us
+917020482655
Need live support?
sales@realtimedatastats.com
Office address
IT Square, Hinjewadi, Pune - 511057 (Maharashtra- India)
Copyright ©2026 Real Time Data Stats. All rights reserved.